METTENIUSIIDAE W. Stull, D. E. Soltis et P. S. Soltis
[Metteniusales+Garryidae]
Takht., Divers. Classif. Fl. Pl.: 352. 24 Apr 1997
Schnizlein, Iconogr. Fam. Regni Veg. 2: ad t. 142*. 1860-1870
Emmotaceae van Tieghem in Bot. Jahresber. (Just) 25(2): 406. 19 Jan 1900;
Emmotales Doweld, Tent. Syst. Pl. Vasc.: li. 23 Dec 2001
Genera/species
11/61
Distribution
Tropical regions in the Old World, Mexico, Central America, the West Indies, tropical South America.
Fossils
Pollen fossils attributed to Platea are known from the Paleocene of North America and Greenland.
Habit
Usually bisexual (Platea dioecious), evergreen trees, shrubs or lianas.
Vegetative anatomy
Phellogen ab initio superficial? Secondary lateral growth in Rhaphiostylis anomalous via successive (concentric) cambia. Vessel elements with simple (Rhaphiostylis) or scalariform (rarely reticulate) perforation plates; lateral pits usually scalariform to opposite (sometimes alternate), bordered or (in at least Poraqueiba and Metteniusa) simple pits. Imperforate tracheary xylem elements tracheids (or fibre-tracheids?) with bordered pits, non-septate. Wood rays uniseriate or multiseriate, homocellular or heterocellular. Axial parenchyma apotracheal usually diffuse-in-aggregates (rarely diffuse), or paratracheal scanty or banded. Tyloses usually absent (sclerotic sometimes abundant). Sieve tube plastids S type? Nodes 1:1, unilacunar with one leaf trace, or 3:3, trilacunar with three traces (in Metteniusa 5:5, pentalacunar with five leaf traces, at petiole base in a U-shaped configuration, distally separate). Heartwood often with gum-like substances. Prismatic calciumoxalate crystals abundant (rhombic crystals abundant in Rhaphiostylis); druses frequent in flowers; styloid-like crystals and crystal groups present in wood ray cells of Apodytes.
Trichomes
Hairs unicellular, unbranched, T-shaped, or absent (in Platea glandular and stellate-peltate or lepidote, sometimes caducous or absent).
Leaves
Alternate (spiral or distichous), simple, entire, in Platea with conduplicate ptyxis. Stipules and leaf sheath absent. Petiole vascular bundle transection arcuate, complex (Metteniusa). Venation pinnate, brochidodromous or eucamptodromous. Stomata anomocytic or cyclocytic (anomocyclocytic). Cuticular wax crystalloids? Mesophyll with fibres. Domatia absent. Leaf margin serrate to entire.
Inflorescence
Terminal (Apodytes) or axillary, panicle, cymose, cymose thyrsoid, fasciculate to corymbose cyme, or compound or simple racemes. Floral prophylls (bracteoles) (two or) three per flower, thick, triangular.
Flowers
Actinomorphic, small to large. Pedicel articulated. Hypogyny. Sepals four or five, with imbricate quincuncial to imbricate aestivation, persistent, connate. Petals four or five, with valvate aestivation, free or connate at base (in Metteniusa late sympetaly, i.e. in late stage connate in lower part; petals absent in female flowers of Platea), sometimes with adaxial hairs. Nectary absent. Disc absent. Secretory hairs present.
Androecium
Stamens four or five, haplostemonous, antesepalous, alternipetalous. Filaments long, subulate, free from each other, free from or adnate (at least at base) to corolla tube (epipetalous). Anthers basifixed to dorsifixed, versatile, usually tetrasporangiate (in Metteniusa polysporangiate; in Emmotum disporangiate, monothecal), septate, latrorse to extrorse or introrse, longicidal (dehiscing by longitudinal slits, in Metteniusa long and moniliform, with 17 or fewer partial microsporangia – locelli – in four vertical rows opening individually); thecae in Metteniusa strongly recurved following dehiscence; connective often enlarged (in Metteniusa massive). Tapetum secretory. Staminodia absent.
Pollen grains
Microsporogenesis simultaneous? Pollen grains tricolporate, shed as monads, bicellular at dispersal? Exine scabrate (Metteniusa) or semitectate, with columellate? infratectum, foveate-reticulate (Apodytes), with ? infratectum, sculpturing?
Gynoecium
Pistil composed of usually three (in Metteniusa five paracarpous) connate carpels, with a single fertile carpel, leading to pseudomonomery (one carpel in Apodytes modified into fleshy dispersal structure; in Emmotum three fertile and two sterile carpels); carpel initials congenitally fused by their margins; gynoecium zygomorphic throughout ontogeny (indicating pseudomonomery). Ovary superior, usually unilocular due to pseudomonomery (in Emmotum bilocular or trilocular, pseudotrimerous), with or without a fleshy appendage. Style single, simple, long, narrow (in Metteniusa filiform), often short. Stigma subcapitate, capitate, punctate, discoid, bilobate or slightly trifid, slightly papillate, type? Pistillodium absent.
Ovules
Placentation apical, subapical to axile (pseudoparietal), collateral or superposed (in Metteniusa parietal). Ovules two per fertile carpel or two per ovary (in Metteniusa one ovule primordium developing in each of two smaller carpels), orthotropous, pendulous, bitegmic (Emmotum) or unitegmic, crassinucellar, anatropous, pendulous, unitegmic, tenuinucellar, three larger carpels (including median carpel) sterile. Integument in Metteniusa more than 20 cell layers thick, massive, highly vascularized. Megagametophyte monosporous, Polygonum type? Endosperm development? Endosperm haustoria? Embryogenesis?
Fruit
A usually one-seeded drupe (with one ovule aborted), often flattened. Pyrene usually asymmetrical, often with ribs or ridges (in Metteniusa asymmetrically arranged) and persistent lateral-basal style.
Seeds
Aril absent. Funicle massive and elongated. Testa membranous, vascularized. Exotestal cells? Endotesta? Tanniniferous cells abundant. Perisperm not developed. Endosperm copious, oily? Embryo usually straight (in Metteniusa curved), short to long, well differentiated, with chlorophyll? Cotyledons two, foliaceous. Germination?
Cytology
n = 12 (Apodytes), 14 (Calatola), 20 (Rhaphiostylis), 22?
DNA
Phytochemistry
Route I iridoids (e.g. daphylloside), monoterpene secoiridoids and emmotins (eudesmane sesquiterpenes), oleanolic acid. Cyanogenic compounds? Tannins present. Aluminium accumulated in Platea.
Use
Edible seeds (Metteniusa edulis), fruits (Poraqueiba).
Systematics
Plateoideae G. W. Stull in Amer. J. Bot. 102: 1809. Nov 2015. Platea (5; P. bullata, P. excelsa, P. latifolia, P. parvifolia, P. sclerophylla; Indochina, Malesia to New Guinea), Calatola (5; C. costaricensis, C. laevigata, C. mollis, C. sanguininensis, C. uxpanapensis; Mexico to Ecuador). – Apodytoideae G. W. Stull in Amer. J. Bot. 102: 1809. Nov 2015. Apodytes (8; tropical and subtropical regions of eastern and southern Africa, Madagascar, southern India, Sri Lanka, Mauritius, Indochina, Malesia to New Guinea, Queensland, New Caledonia), Rhaphiostylis (11; tropical West Africa), Dendrobangia (2; D. boliviana, D. multinervia; Costa Rica, tropical South America). – Metteniusoideae G. W. Stull in Amer. J. Bot. 102: 1809. Nov 2015. Emmotum (12; tropical South America), Poraqueiba (3; P. guianensis, P. paraensis, P. sericea; tropical South America); Oecopetalum (3; O. greenmanii, O. guatemalense, O. mexicanum; Mexico, Central America), Pittosporopsis (1; P. kerrii; Southeast Asia), Metteniusa (7; M. cogolloi, M. cundinamarcensis, M. edulis, M. huilensis, M. nucifera, M. santanderensis, M. tessmanniana; Costa Rica, Panamá, the northern Andes to Peru and western Venezuela), Ottoschulzia (4; O. cubensis, O. domingensis, O. pallida, O. rhodoxylon; Guatemala, the West Indies).
Calatola has vessel elements with scalariform perforation plates, nodes 3:3, trilacunar with three leaf traces, leaves with conduplicate ptyxis and serrate leaf margin.
Rhaphiostylis is very similar to Icacinaceae in its anatomy, with the exception of the banded parenchyma present in Icacinaceae.
Dendrobangia has solitary vessels with scalariform perforation plates with numerous bars and the lateral (intervessel) pits are opposite. The fibres have distinctly bordered pits. The axial parenchyma is usually diffuse-in-aggregates and scanty paratracheal. The axial parenchyma cells are chambered and contain prismatic crystals. Uniseriate and 8-seriate rays are present and the body cells are procumbent and square with few small prismatic crystals and small styloids. The leaves have lepidote-stellate hairs, brochidodromous leaf venation and cyclocytic stomata. The inflorescence is an axillary panicle. The bracteoles are two to six. The five sepals are imbricate and the five petals are basally fused into a tube and each lobe has a strap-like, apically broadened acumen. The five stamens are adnate to the corolla tube and alternate to the lobes. The anthers are basifixed. The ovary is unilocular and uniovulate. The fruit is a one-seeded drupe. The embryo is minute with a copious endosperm.
Metteniusa forms a weakly supported clade together with [Oecopetalum+Ottoschulzia] in analyses by Byng & al. (2014). Emmotum and Ottoschulzia possess vessel elements with scalariform perforation plates with less than eleven cross-ribs.
Emmotum is somewhat similar to Oncotheca (Oncothecaceae) in its monosporangiate thecae and in the gynoecium having both fertile and sterile carpels.
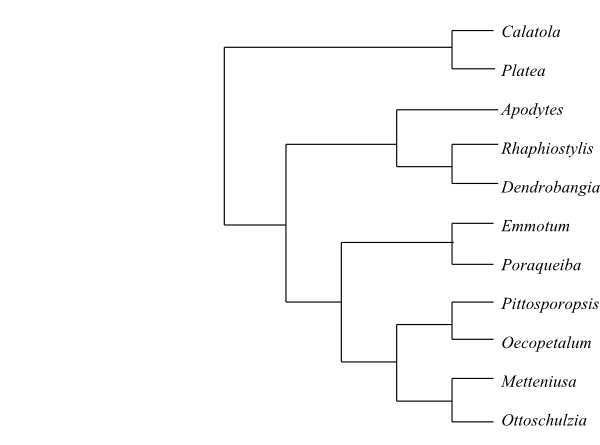 |
|
Cladogram of
Metteniusaceae
based on DNA sequence data (Stull & al. 2015).
|
Literature
Baas P. 1973. Epidermal leaf
characters of the Malesian Icacinaceae. – Acta Bot. Neerl. 22:
329-359.
Baas P. 1974. Stomatal types in
Icacinaceae. Additional observations on genera outside Malesia. –
Acta Bot. Neerl. 23: 193-200.
Bailey IW, Howard RA. 1941a.
The comparative morphology of the Icacinaceae I. Anatomy of the
node and internode. – J. Arnold Arbor. 22: 125-132.
Bailey IW, Howard RA. 1941b.
The comparative morphology of the Icacinaceae II. Vessels. – J.
Arnold Arbor. 22: 171-187.
Bailey IW, Howard RA. 1941c.
The comparative morphology of the Icacinaceae III. Imperfect
tracheary elements and xylem parenchyma. – J. Arnold Arbor. 22:
432-442.
Bailey IW, Howard RA. 1941d.
The comparative morphology of the Icacinaceae IV. Rays of the
secondary xylem. – J. Arnold Arbor. 22: 556-568.
Bâthie HP de la. 1952.
Icacinacées (Icacinaceae). – In: Humbert H (ed), Flore de
Madagascar et des Comores 119, Typographie Firmin-Didot et
Cie, Paris.
Bautista PB, De Andrade TAP.
1975. O pólen em plantas da Amazônia V. Contribuição ao estudo da
familia Icacinaceae. – Bol. Mus. Paraense “Emilio Goeldi”, N. S.,
Bot. 47: 1-11.
Braga de Oliveira A. 1995.
Terpenoids from Amazonian Icacinaceae. – In: Chemistry of the
Amazon, ACS Symposium Ser., Vol. 588, pp. 99-115.
Byng JW, Bernardini B, Joseph
JA, Chase MW, Utteridge TMA. 2014. Phylogenetic relationships of
Icacinaceae focusing on the vining genera. – Bot. J. Linn. Soc.
176: 277-294.
Capuron R. 1970. Notes sur les
Icacinaceae. – Adansonia, n. s., 10: 507-510.
Dahl AO. 1952. The comparative
morphology of the Icacinaceae VI. The pollen. – J. Arnold Arbor.
33: 252-295.
Dahl AO. 1955. The pollen
morphology of several genera excluded from the family
Icacinaceae. – J. Arnold Arbor. 36: 159-163.
Duno De Stefano R. 2007.
Tratamiento taxonómico del género Dendrobangia Rusby
(Cardiopteridaceae o Icacinaceae). – Candollea 62: 91-103.
Duno De Stefano R,
Fernández-Concha GC. 2011. Morphology-inferred phylogeny and a
revision of the genus Emmotum (Icacinaceae). – Ann.
Missouri Bot. Gard. 98: 1-27.
Duno De Stefano R. Angulo DF, Stauffer FW. 2007. Emmotum harleyi, a new species from Bahia, Brazil, and lectotypification of other Icacinaceae. – Novon 17: 306-309.
Endress PK, Rapini A. 2014.
Floral structure of Emmotum (Icacinaceae sensu stricto
or Emmotaceae), a phylogenetically isolated genus of lamiids with
a unique pseudotrimerous gynoecium, bitegmic ovules and
monosporangiate thecae. – Ann. Bot. 114: 945-959.
Engler A. 1896. Icacinaceae. –
In: Engler A, Prantl K (eds), Die natürlichen Pflanzenfamilien
III(5), W. Engelmann, Leipzig, pp. 233-257, 459-460; Engler A.
1897. Nachträge zu III(5), pp. 225-227.
Fagerlind F. 1945. Bau des
Gynöceums, der Samenanlage, und des Embryosackes bei einigen
Repräsentanten der Familie Icacinaceae. – Svensk Bot. Tidskr. 39:
346-364.
González FA, Rudall PJ. 2010.
Flower and fruit characters in the early-divergent lamiid family
Metteniusaceae, with particular reference to the evolution of
pseudomonomery. – Amer. J. Bot. 97: 191-206.
González FA, Betancur J, Maurin
O, Freudenstein JV, Chase MW. 2007. Metteniusaceae, an
early-diverging family in the lamiid clade. – Taxon 56:
795-800.
Guymer GP. 1984. Icacinaceae. –
In: George AS (ed), Flora of Australia 22, Australian Government
Publ. Service, Canberra, pp. 204-212.
Haron NW, Ping ST. 1997.
Distribution and taxonomic significance of flavonoids in the
Olacaceae and Icacinaceae. – Biochem. Syst. Ecol. 25:
265-263.
Heintzelmann CE, Howard RA.
1948. The comparative morphology of the Icacinaceae V. The
pubescence and the crystals. – Amer. J. Bot. 35: 42-52.
Howard RA. 1940. Studies of the
Icacinaceae I. Preliminary taxonomic notes. – J. Arnold Arbor.
21: 461-489.
Howard RA. 1942a. Studies of
the Icacinaceae II. Humirianthera, Leretia,
Mappia and Nothapodytes, valid genera of the
Icacineae. – J. Arnold Arbor. 23: 55-78.
Howard RA. 1942b. Studies of
the Icacinaceae III. A revision of Emmotum. – J. Arnold
Arbor. 23: 479-494.
Howard RA. 1942c. Studies of
the Icacinaceae IV. Considerations of the New World genera. –
Contr. Gray Herb. Harvard Univ. 142: 3-60.
Howard RA. 1943. Studies of the
Icacinaceae VIII. Brief notes of some Old World genera. – Lloydia
6: 144-154.
Kaplan MAC, Ribeiro J, Gottlieb
OR. 1991. Chemographical evolution of terpenoids in Icacinaceae.
– Phytochemistry 30: 2672-2676.
Kårehed J. 2001. Multiple
origin of the tropical forest tree family Icacinaceae. – Amer. J.
Bot. 88: 2259-2274.
Kårehed J. 2002. Not just
hollies – the expansion of Aquifoliales. – In: Evolutionary
studies in asterids emphasising euasterids II, Ph.D. diss., Acta
Universitatis Upsaliensis, Uppsala, Sweden, pp. 1-14.
Karsten H. 1859.
Metteniusa Karst. – Flora of Colombia 1: 79-80, t.
39.
Kronfeld M. 1896. Aquifoliaceae. – In:
Engler A, Prantl K (eds), Die natürlichen Pflanzenfamilien
III(5), W. Engelmann, Leipzig, pp. 183-189.
Labat J-N, Rabevohitra R,
El-Achkar E. 2006. Révision synoptique du genre Apodytes
(Icacinaceae) à Madagascar et aux Comores. – Adansonia, sér. III,
28: 379-387.
Lozano-Contreras G, de Lozano
NB. 1988. Metteniusaceae. – In: Pinto P, Lozano-Contreras G
(eds), Flora de Colombia, Monografia 11, Instituto de Ciencias
Naturales, Universidad Nacional de Colombia, Bogotá, pp.
1-53.
Lozano-Contreras G, Lozano NB
de. 1994. Una nueva especie de Metteniusa Karsten
(Metteniusaceae) de Colombia. – Novon 4: 266-270.
Lucas GL. 1968. Icacinaceae. –
In: Milne-Redhead E, Polhill RM (eds), Flora of tropical East
Africa, Crown Agents for Overseas Governments and
Administrations, London, pp. 1-18.
Mauritzon J. 1936.
Embryologische Angaben über Stackhousiaceae, Hippocrateaceae und
Icacinaceae. – Svensk Bot. Tidskr. 30: 541-550.
Mendes EJ. 1963. 50.
Icacinaceae. – In: Exell AW, Fernandes A, Wild H (eds), Flora
Zambesiaca 2 (Part 1), Crown Agents for Oversea Governments and
Administrations, London, pp. 340-351.
Miers J. 1852.
Observations on the affinities of the
Icacinaceae.
– Ann. Mag. Nat. Hist. 2nd ser. 9: 218-226.
Perrier de la Bâthie H. 1944.
Révision des Icacinacées de Madagascar et des Comores. – Mém.
Mus. Natl. Hist. Nat. 18: 289-308.
Pigg KB, Manchester SR, DeVore
ML. 2008. Fruits of Icacinaceae (tribe Iodeae) from the Late
Paleocene of western North America. – Amer. J. Bot. 95:
824-832.
Pittier H. 1925. Árboles y
arbustos nuevos de Venezuela. – Bol. Sci. Técn. Mus. Comercial
Venezuela 1: 45-47.
Potgieter MJ, Wyk AE van.
1994b. Two new species of Apodytes (Icacinaceae) from
southern Africa. – South Afr. J. Bot. 60: 231-239.
Potgieter MJ, Wyk AE van.
1994c. Fruit structure of the southern African species of
Apodytes E. Meyer ex Arn. (Icacinaceae). – Bot. J. Linn.
Soc. 115: 221-233.
Potgieter MJ, Wyk AE van. 1999.
Leaf anatomy of the southern African Icacinaceae and its
taxonomic significance. – South Afr. J. Bot. 65: 153-162.
Rankin BD, Stockey RA, Beard G.
2008. Fruits of Icacinaceae from the Eocene Appian Way locality
of Vancouver Island, British Columbia. – Intern. J. Plant Sci.
169: 305-314.
Shweta S, Zuehlke S, Ramesha
BT, Priti V, Kumar PM, Ravikanth G, Spiteller M, Vasudeva R,
Shaanker RU. 2010. Endophytic fungal strains of Fusarium
solani, from Apodytes dimidiata E. Mey. ex Arn.
(Icacinaceae) produce camptothecin, 10-hydroycamptothecin and
9-methoxycamptothecin. – Phytochemistry 71: 117-122.
Sleumer H. 1934. Eine neue Art
der Gattung Aveledoa Pittier. – Notizbl. Bot. Gart.
Berlin-Dahlem 12: 148-150.
Sleumer H. 1936. Über die
Gattung Metteniusa Karsten (=Aveledoa Pittier).
– Notizbl. Bot. Gart. Berlin-Dahlem 13: 359-361.
Sleumer H. 1942. Icacinaceae. –
In: Engler A (†), Harms H, Mattfeld J (eds), Die natürlichen
Pflanzenfamilien, 2. Aufl., Bd. 20b, W. Engelmann, Leipzig, pp.
322-396.
Sleumer H. 1969. Materials
towards the knowledge of the Icacinaceae of Asia, Malesia, and
adjacent areas. – Blumea 17: 181-264.
Sleumer H. 1971. Icacinaceae. –
In: Steenis CGGJ van (ed), Flora Malesiana I, 7,
Wolters-Noordhoff, Groningen, pp. 1-87.
Spichiger R, Savolainen V,
Manen J-F. 1993. Systematic affinities of Aquifoliaceae and
Icacinaceae from molecular data analysis. – Candollea 48:
459-464.
Staveren MGC van, Baas P. 1973.
Epidermal leaf characters of Malesian Icacinaceae. – Acta Bot.
Neerl. 22: 329-359.
Stull GW, Moore RW, Manchester
SR. 2011. Fruits of Icacinaceae from the Eocene of southeastern
North America and their biogeographic implications. – Intern. J.
Plant Sci. 172: 935-947.
Stull GW, Stefano RD de, Soltis DE, Soltis PS. 2015. Resolving basal lamiid phylogeny and the circumscription of Icacinaceae with a plastome-scale data set. – Am. J.
Bot. 102: 1794-1813.
Teo SP, Haron NW. 1999.
Anatomical studies in West Malaysian Icacinaceae. – Aust. Syst.
Bot. 11: 729-738.
Utteridge TMA. 2010. A new
species of Platea (Icacinaceae) from Peninsular
Malaysia: Platea malayana. – Kew Bull. 65: 345-348.
Villiers J-F. 1973.
Icacinaceae. – In: Aubréville A, Leroy J-F (eds), Flore de Gabon
20, Muséum National d’Histoire Naturelle, Paris, pp. 3-100.
Villier J-F. 1980. Icacinaceae.
– In: Aubréville A, Leroy J-F (eds), Flore de la Nouvelle
Calédonie 9, Muséum National d’Histoire Naturelle (Paris), pp.
159-174.
Wagner R. 1923. Über
Vorkommnisse von Domatien bei Icacinaceen. – Anz. Akad. Wiss.
Wien., Math.-Naturw. Kl. 60: 189-193.
